Score calculation and percent risk of conversion to secondary-progressive multiple sclerosis over five years. Intended for use in relapsing-remitting multiple sclerosis patients.
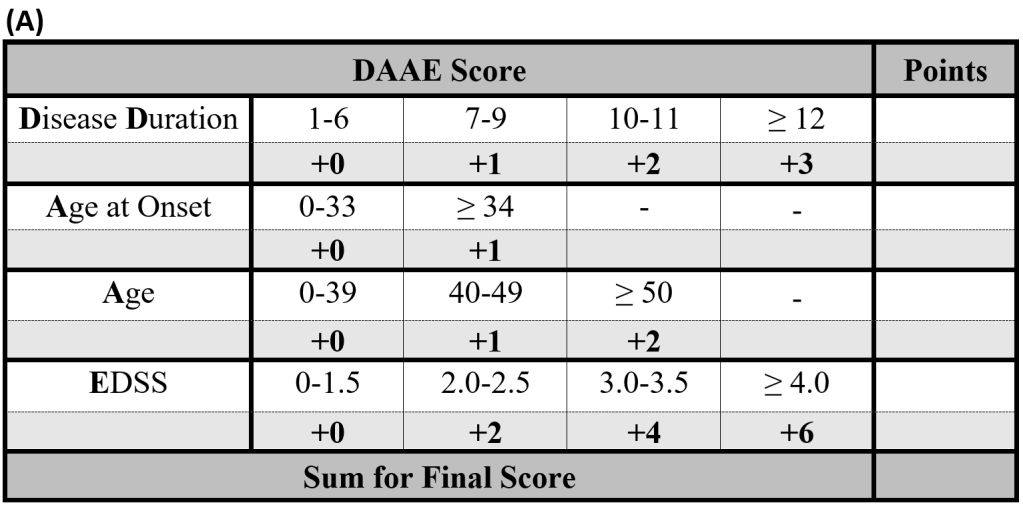
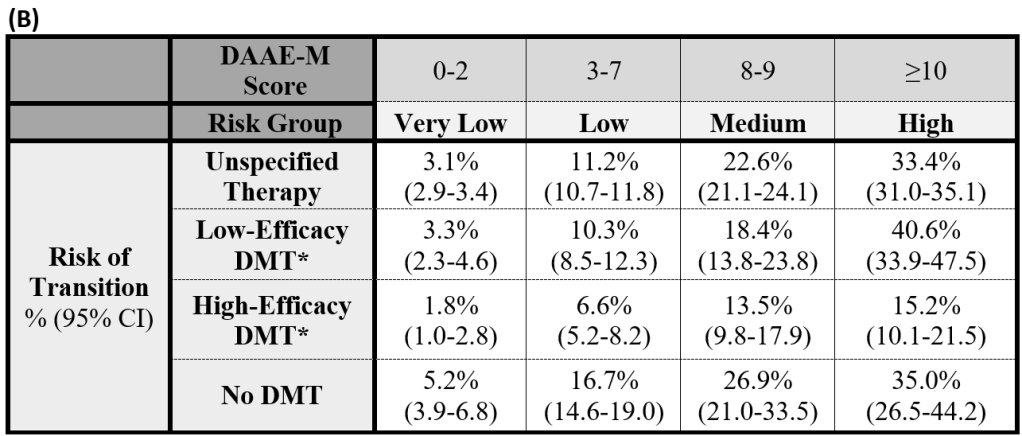
To calculate the DAAE Score, determine the points scored for each factor in Table (A). The total sum of these points is the DAAE Score. Reference the DAAE Score with risk group and percent risk of conversion in Table (B).
Abbreviations: EDSS=expanded disability status scale; DMT=disease modifying therapy; CI=confidence interval
*DMT is presumed to be used over the majority (>50%) of the five-year period. Unspecified therapy relates to estimated risks without accounting for DMT.
BACKGROUND INFORMATION
The DAAE-M Score integrates Disease duration, Age at onset, Age, Expanded Disability Status Scale, and disease modifying therapy to estimate longitudinal risk.
DMT efficacy scheme: High – rituximab, ocrelizumab, mitoxantrone, alemtuzumab, natalizumab, ofatumumab; Low – interferon-beta, glatiramer acetate, teriflunomide, dimethyl fumarate
This work was supported by the European Committee for Treatment and Research in Multiple Sclerosis. Unspecified therapy estimates are based on international combined data from the United States (Jacobs Multiple Sclerosis Center, n=1,309), Netherlands (Multiple Sclerosis Center Amsterdam, n=877), and the MSBase multi-center international dataset (n=34,510). DMT efficacy DMT risk estimates are based on a propensity-score-matched data subset from the MSBase registry (n=9,547).
For questions, contact Tom A Fuchs, t.fuchs@amsterdamumc.nl
Preliminary DAAE Score development manuscript

Leave a comment